About the haber process

Old equipment of Haber Process
What is the Haber Process?
The Haber process is used to manufacture ammonia from nitrogen, which made 80% of the air and hydrogen, derived mainly from natural gas Ammonia. The overall equation of the Haber process is N2(g) + 3H2(g) <--> 2NH3(g) ΔH = −92.22 kJ·mol−1, which is a reversible and exothermic reaction. Ammonia could then be used to make nitric acid, which reacts with ammonia to create ammonium nitrate, which is a very important fertilizer. In the old days, Fritz Haber used a very simple equipment to test the Haber Process for multiple times. This equipment is shown on the left.
The Haber process is used to manufacture ammonia from nitrogen, which made 80% of the air and hydrogen, derived mainly from natural gas Ammonia. The overall equation of the Haber process is N2(g) + 3H2(g) <--> 2NH3(g) ΔH = −92.22 kJ·mol−1, which is a reversible and exothermic reaction. Ammonia could then be used to make nitric acid, which reacts with ammonia to create ammonium nitrate, which is a very important fertilizer. In the old days, Fritz Haber used a very simple equipment to test the Haber Process for multiple times. This equipment is shown on the left.
---------------------------------------------------------------------------------------------------------
CLICK TO ENLARGE
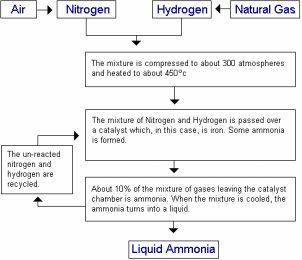
The process of Haber Process
Raw materials for creating ammonia are air for nitrogen (N2(g)) and methane and water for hydrogen (H2(g)). Hydrogen is process by taking methane (CH4(g)) and reacting it with steam (H2O(g)) and thus creating carbon dioxide (CO2(g)) and hydrogen (H2(g)). The nitrogen (N2(g)) is obtained from the air by fractional distillation, because the air is made up of 80% nitrogen. Fractional distillation is where they take a substance with another substance mixed together, and since both substances have different boiling points, they heat up the mixture. When the one substance with the lowest boiling point starts to boil it evaporates into a cooling jacket, which then liquidifies and is poured into a beaker or container. Then the next substance starts to boil and does the same thing except the substance is put into a different beaker or container to store it. And this separates all of the components of mixtures and you can get one or more pure substances out of a mixture.
Gaining the raw materials of the Haber Process, these raw materials will go through the process beside and formed a liquid Ammonia. Click the image to enlarge it.
---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Below is video of the Haber Process !!! Enjoy !!!
Raw materials for creating ammonia are air for nitrogen (N2(g)) and methane and water for hydrogen (H2(g)). Hydrogen is process by taking methane (CH4(g)) and reacting it with steam (H2O(g)) and thus creating carbon dioxide (CO2(g)) and hydrogen (H2(g)). The nitrogen (N2(g)) is obtained from the air by fractional distillation, because the air is made up of 80% nitrogen. Fractional distillation is where they take a substance with another substance mixed together, and since both substances have different boiling points, they heat up the mixture. When the one substance with the lowest boiling point starts to boil it evaporates into a cooling jacket, which then liquidifies and is poured into a beaker or container. Then the next substance starts to boil and does the same thing except the substance is put into a different beaker or container to store it. And this separates all of the components of mixtures and you can get one or more pure substances out of a mixture.
Gaining the raw materials of the Haber Process, these raw materials will go through the process beside and formed a liquid Ammonia. Click the image to enlarge it.
---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Below is video of the Haber Process !!! Enjoy !!!
